| 所属 |
① 九州大学 大学院薬学研究院 ② 九州大学 大学院薬学研究院 |
|
|---|---|---|
| 氏名 |
① CAAVEIRO Jose ② 植田 正 |
|
| AMED 事業 |
課題名 | グリーンファルマ創薬構造解析による支援高度化の推進 |
| 代表機関 | 九州大学 | |
| 代表者 | 大戸 茂弘 | |
膜蛋白質、ナノディスク、構造生物学、クライオ電子顕微鏡、相互作用解析
Membrane proteins are at the forefront of Pharmaceutical research. These group of proteins continue to be critical targets for numerous therapeutic approaches in human disease. However, membrane proteins are notoriously difficult to obtain and purify partly because they are separated from their natural milieu, the membrane, and transferred to detergent micelles which similar but not identical properties to lipid bilayers.
Nanodiscs are particles of nanometer scale composed of a belt-protein (apolipoprotein), and lipids of the same class as those present in biological membranes. Therefore, nanodiscs better mimic the natural environment of the plasma membrane (Figure 1, 2).
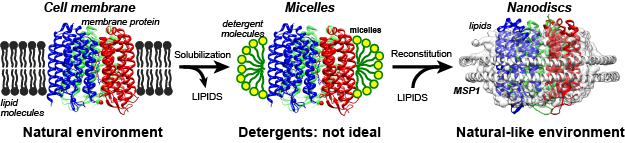
Figure 1. Overview of nanodiscs.
We employ nanodiscs as a tool to study membrane proteins in pharmaceutical research, such as for compound screening, interaction analysis, and structural characterization such as Cryo-EM.
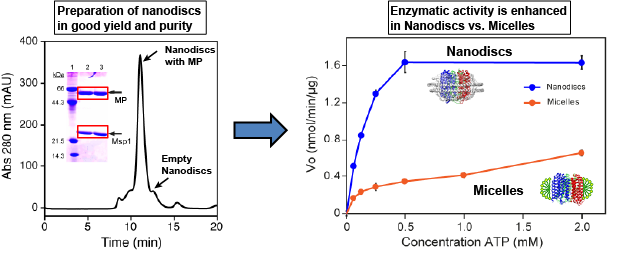
Figure 2. Nanodiscs better preserve the enzymatic activity of a membrane protein (MP) compared with micelles.
Nanodiscs are employed for various purposes in pharmaceutical and basic research such as the following examples.
(1)Enzymatic activity. Nanodiscs are superior to preserve the activity of membrane proteins.
(2)Compound screening. If sufficient amount of nanodiscs are obtained, membrane protein can be subjected to various screening procedures.
(3)Structural studies. Because of their size and stability, nanodiscs are ideal for the structural characterization of membrane proteins by the technique of Cryo-EM. In some favorable cases, the binding of small compounds and drug candidates can be observed, facilitating the development of molecules of greater potency by structure-based design (Figure 3).
(4)Interaction analysis. Nanodiscs are employed to study the direct interaction of membrane proteins with natural ligands, drug candidates, or other partner proteins and biomolecules. If certain conditions meet, such as sufficient protein yield, high-resolution biophysical techniques may be employed.
The preparation of membrane proteins in nanodiscs is summarized in Figure 1 and Figure 4. Basically, membrane proteins in detergent are incubated with optimized concentrations of lipids and apolipoprotein, leading to the formation of nanodiscs upon removal of detergent. Nanodiscs are further purified by gel filtration and/or affinity chromatography.
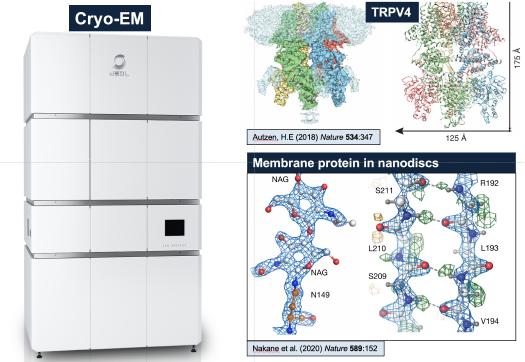
Figure 3. Nanodiscs are ideally suited for structural analysis by Cryo-EM.
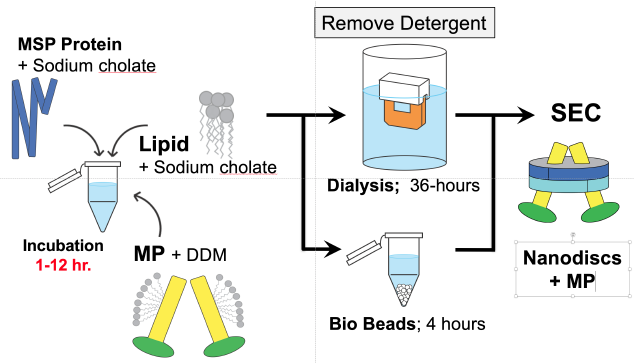
Figure 4. Outline of preparation of nanodiscs (MP, membrane protein; DDM, detergent dodecyl maltoside; MSP, apolipoprotein; SEC, size exclusion chromatography)
Our research is similar to the outline exposed above. We are interested to discover new molecules with therapeutic potential using membrane proteins as targets, and nanodiscs are an essential part of our research. We also employ nanodiscs to advance the basic understanding of membrane proteins, such as their structure, stability and interaction with other partner biomolecules using high-resolution biophysical techniques.